TEST DIRECTIONS FOR MENOPAUSE (FSH) TESTS
WHEN TO START TESTING
If the subject is still having monthly periods, then the first test should be taken during the first week of her menstrual cycle (Days 2-7, with Day 1 being the first day of menstruation). Repeat with the second test 1 week later.
If the subject is no longer having regular periods, the first test should be taken at any time during the month and the second test should be taken 1 week later.
DIRECTIONS FOR USE
Allow Test Device & Urine Specimen to reach room temperature (15-300C) prior to testing.
1. Determine the day you will begin testing. (See the above section: “WHEN TO START TESTING”).
2. Remove the test device from the sealed pouch and use it as soon as possible.
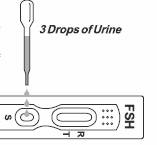
3. Place the test device on a clean and level surface. Hold the dropper vertically and transfer 3 full drops of urine (approx. 100μL) to the specimen well (S) of the test device, and then start the timer. Avoid trapping air bubbles in the specimen well (S). See illustration below.
4. Start the timer and wait for the red line(s) to appear. The result should be read
at 5 minutes. Do not interpret the result after 10 minutes.
SAMPLE TEST & RESULTS:
|
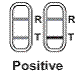
POSITIVE: Two lines are visible and the color intensity of A positive result means that the FSH level is
|
|
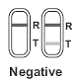
NEGATIVE: Two lines are visible, but the line in the Test A negative result means that the FSH level is not |
|
INVALID: If there is no line in the Reference Line Region The most likely reasons for an invalid result are |
TEST INTERPRETATION:
1st Test |
2nd Test |
Interpretation |
Positive |
Positive |
Most likely in perimenopause. |
Positive Negative |
May be in early stages of |
|
Negative |
Negative |
Most likely not experiencing |
LIMITATIONS:
- The One Step Test Device provides only a qualitative, preliminary analytical result. A secondary analytical method must be used to obtain a confirmed result. Gas Chromatography/Mass Spectrometry (GC/MS) are the preferred confirmatory methods.
- It is possible that technical or procedural errors, as well as other interfering substances in the urine specimen may cause erroneous results.
- A Positive Result indicates presence of the drug or its metabolites but does not indicate level of intoxication, route of administration or concentration in urine.
- A Negative Result may not necessarily indicate drug-free urine. Negative Results may be obtained when drug is present but below the cutoff level of the test.
Disclaimer: DrugTestStrips.com products have been found to be highly accurate and are guaranteed against defects in labeling & packaging until the stated expiration date. Regardless, DrugTestStrips.com will not be held accountable/liable for the outcome of ANY result arising from the use of Drug Test Strips products.
DrugTestStrips.com sole obligation in the event of ANY damage/result/defect is to replace it with a comparable new test strip product. Except for such replacement, the sale, transfer, or handling of these drug test products are without warranty, condition or liability even though the damage/result/defect is due to negligence or other fault.